Cobalt-Doped Bismuth Nanosheet Catalyst for Enhanced Electrochemical CO2 Reduction to Electrolyte-Free Formic acid
Cobalt-Doped Bismuth Nanosheet Catalyst for Enhanced Electrochemical CO2 Reduction to Electrolyte-Free Formic acid
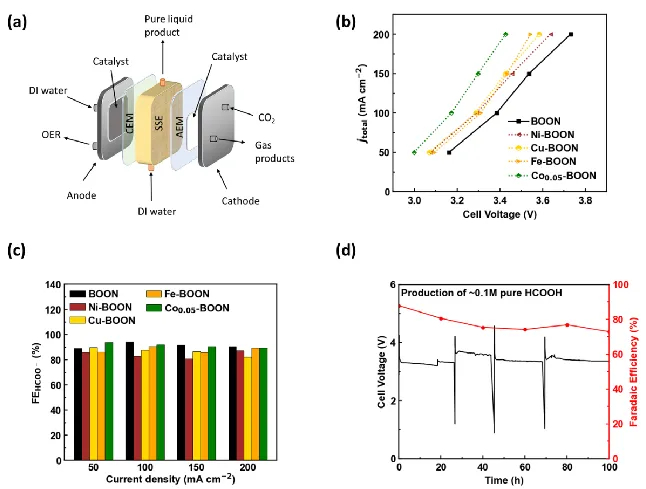
Electrochemical carbon dioxide (CO2) reduction reaction (CO2RR) to valuable liquid fuels, such as formic acid/formate (HCOOH/HCOO−) is a promising strategy for carbon neutrality. Enhancing CO2RR activity while retaining high selectivity is critical for commercialization. To address this, we developed metal-doped bismuth (Bi) nanosheets via a facile hydrolysis method. These doped nanosheets efficiently generated high-purity HCOOH using a porous solid electrolyte (PSE) layer. Among the evaluated metal-doped Bi catalysts, Co-doped Bi demonstrated improved CO2RR performance compared to pristine Bi, achieving ~90% HCOO− selectivity and boosted activity with a low overpotential of ~1.0 V at a current density of 200 mA cm-2. In a solid electrolyte reactor, Co-doped Bi maintained HCOOH Faradaic efficiency of ~72% after a 100-hour operation under a current density of 100 mA cm-2, generating 0.1 M HCOOH at 3.2 V. Density functional theory (DFT) results revealed that Co-doped Bi required a lower applied potential for HCOOH generation from CO2, due to stronger binding energy to the key intermediates OCHO* compared to pure Bi. This study shows that metal doping in Bi nanosheets modifies the chemical composition, element distribution, and morphology, improving CO2RR catalytic activity performance by tuning surface adsorption affinity and reactivity.