Guiding CO2RR Selectivity by Compositional Tuning in the Electrochemical Double Layer
The electrochemical conversion of carbon dioxide to value-added chemicals provides an environmentally benign alternative to current industrial practices. However, current electrocatalytic systems for the CO2 reduction reaction (CO2RR) are not practical for industrialization, owing to poor specific product selectivity and/or limited activity. Interfacial engineering presents a versatile and effective method to direct CO2RR selectivity by fine-tuning the local chemical dynamics.
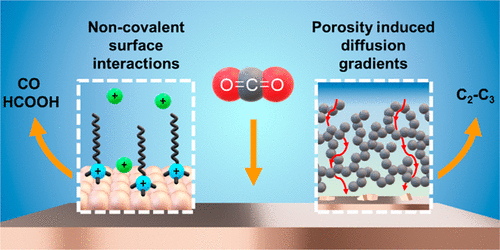
This Account describes interfacial design strategies developed in our laboratory that use electrolyte engineering and porous carbon materials to modify the local composition at the electrode–electrolyte interface.Our first strategy for influencing surface reactivity is to perturb the electrochemical double layer by tuning the electrolyte composition. We approached this investigation by considering how charged molecular additives can organize at the electrode surface and impact CO2 activation. Using a combination of advanced electrochemical techniques and in situ vibrational spectroscopy, we show that the surfactant properties (the identity of the headgroup, alkyl chain length, and concentration) as well as the electrolyte cation identity can affect how surfactant molecules assemble at a biased electrode. The interplay between the electrolyte cations and the surfactant additives can be regulated to favor specific carbon products, such as HCOO–, and suppress the parasitic hydrogen evolution reaction (HER). Together, our findings highlight how molecular assemblies can be used to design selective electrocatalytic systems.
In addition to the electrolyte design, the local spatial confinement of reaction intermediates presents another strategy to direct CO2RR selectivity. We were interested in uncovering the role of porous carbon-supported catalysts toward selective carbon product formation. In our initial study, we show that carbon porosity can be optimized to enhance C2H4 and CO selectivity in a series of Cu catalysts embedded in a tunable carbon aerogel matrix. These results suggested that local confinement of the active surface plays a role in CO2 activation and motivated an investigation into probing how this phenomenon can be translated to a planar Cu electrode. Our findings show that carbon modifiers facilitated surface reconstruction and regulated CO2 diffusion to suppress HER and improve the C2–3 product selectivity. Given the ubiquity of carbon materials in catalysis, this work demonstrates that carbon plays an active role in regulating selectivity by restricting the diffusion of substrate and reaction intermediates. Our work in tuning the composition of the electrochemical double layer for increased CO2RR selectivity demonstrates the potential versatility in boosting catalytic performance across an array of catalytic systems.

I am so grateful for your article post. Really thank you! Really Great. Mac Thie
Regards for all your efforts that you have put in this. very interesting info. Mathilde Paddy Cocks
Very good article. I am experiencing some of these issues as well.. Glen Mullison
Thanks for sharing, this is a fantastic blog article. Really thank you! Really Great. Myles Tramm
Great post. I will be dealing with many of these issues as well.. Daryl Dugger
Anyone who is a hardcore a bit more complicated than that. Herbert Martinov
Awesome! Its truly awesome post, I have got much clear idea concerning from this paragraph. Ervin Portis
Wohh precisely what I was searching for, thank you for posting. Ariel Purkerson
Howdy! I just want to give you a big thumbs up for the great information you have here on this post. I am coming back to your website for more soon. Geoffrey Ayre
Thank you for your article. Thanks Again. Want more. Charles Sofranko
I am sure this article has touched all the internet visitors, its really really good piece of writing on building up new web site. Elvin Trio
I enjoy your writing style truly loving this internet site . Lazaro Goedicke
Wohh exactly what I was searching for, appreciate it for posting. Denver Newhart
I’m really inspired along with your writing talents and also with the structure for your weblog. Is this a paid theme or did you modify it yourself? Either way stay up the excellent high quality writing, it?s uncommon to see a nice blog like this one today..
Keep on working, great job!
This site definitely has all of the information I needed about this subject
Appreciate you sharing, great article.Really thank you! Keep writing.
Major thanks for the article post. Much thanks again.
Loving the information on this website , you have done great job on the blog posts.
Thanks for sharing, this is a fantastic blog post.Really thank you! Much obliged.
My website: порно писсинг
A lot of blog writers nowadays yet just a few have blog posts worth spending time on reviewing.
My website: gayporno.pro
Excellent site. A lot of helpful information here. I?¦m sending it to several pals ans additionally sharing in delicious. And naturally, thanks on your effort!